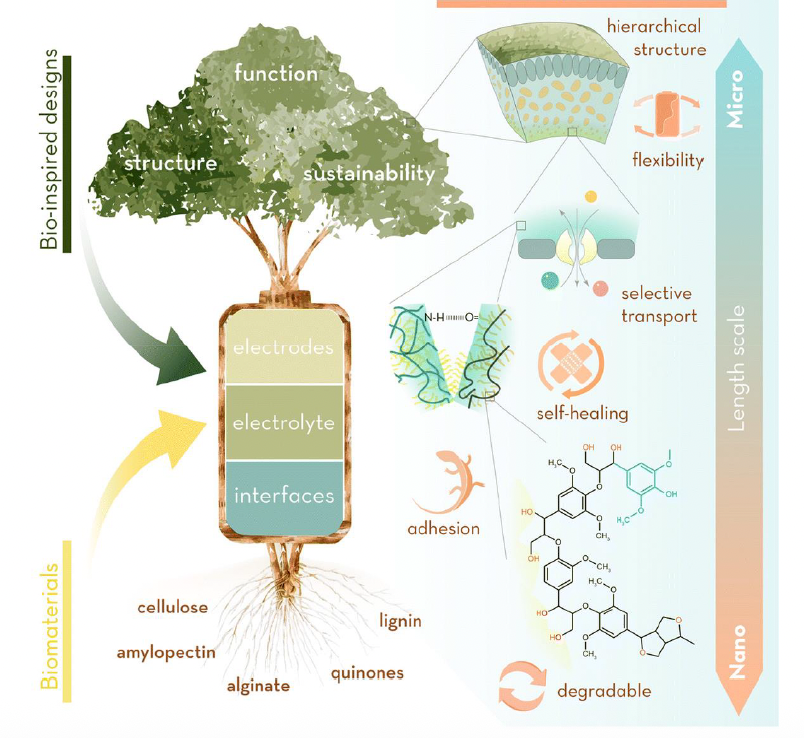
MIT researchers improve our understanding of lithium dendrite formation
Lithium metal electrodes are a key component in modern batteries, but their long-term potential is hampered by the fact that repeated charging will cause lithium filament deposits to creep into the liquid electrolyte. Up until now, researchers have not studied these deposits in detail, but now an MIT research team have released a detailed analysis of their “fundamental experimental and theoretical insights into the growth of lithium metal”.
To study the deposit formation, the team used a glass capillary cell. Their findings, published in the Energy and Environmental Science journal, showed that there are actually two types of deposit, formed under different applied current conditions. ‘Mossy’ deposits are slow growing, and build up from the base as they feed on the electrolyte. ‘Dendritic’ projections are more like fingers, rapidly spiking forward at the tips of the build-up.
Dendritic projections can be more devastating, capable of penetrating the battery separator material and potentially triggering a fire. However, the good news is that they form only under high-current conditions which should not occur in the majority of batteries. Because dendritic projections do not form in normal conditions, previous research had not reliably distinguished them from the more common mossy buildup.
As for these mossy deposits, the researchers found that their growth can be blocked by a hard nanoporous ceramic layer between electrode and electrolyte. This gives battery designers a safe current range, within which dendritic lithium will not form, and mossy deposition can be suppressed with a physical barrier.
To read the full details of the MIT paper, follow the link here.